As our clinics grow busier and our imaging options expand, we must balance our desire to learn as much as we can about a patient’s anatomy with our requirement to efficiently manage cases. When it comes to imaging, I prioritize speed and accuracy; a test that takes too long to process has the potential to disrupt my clinic, and a test that yields unreliable results is useless.
OCT angiography (OCTA) has become an increasingly important tool in diagnosis and monitoring of diabetic eye disease and uveitis. I have begun to use, among my arsenal of imaging platforms, HD AngioPlex software on the ZEISS CIRRUS 6000 100 kHz spectral-domain OCT/OCTA.
In this piece, I illustrate how new imaging technology informs my clinic’s efficiency.
DIABETIC EYE DISEASE
Before ordering imaging tests for patients with diabetic eye disease, I ask myself if the results will affect my management of the case. In cases of diabetic eye disease with the presence of macular edema and/or leakage due to neovascularization, I often order OCT imaging with wide-field fluorescein angiography (FA). I believe imaging of the peripheral retina and assessing the peripheral perfusion and vascular leakage can be useful. At that time, OCTA imaging did not rapidly image the peripheral retina, generate a reproducible readout, or provide measurement data by which to predict disease progression and treatment outcomes.
Recent imaging advances, however, may change the way I practice. Fast and reproducible results allow me to quickly assess the peripheral retina, the patient’s diffusion status, and easily identify neovascularization.
Take, for example, the AngioPlex HD software. The platform’s 6x6 scans provide 96% more data than standard AngioPlex scans of the same size; for 8x8 scans, the new platform provides 240% more data (Figure 1).
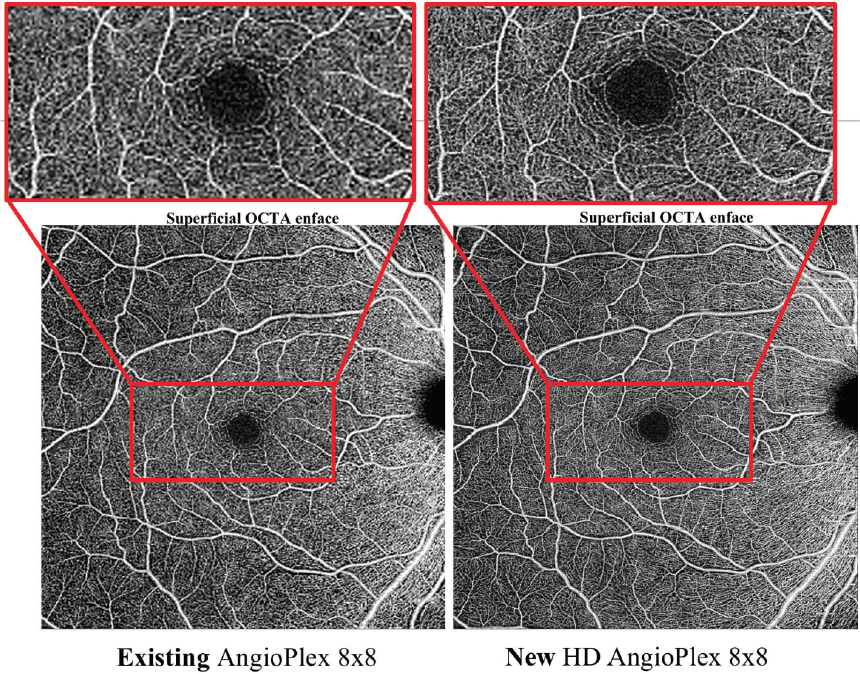
Figure 1. On the AngioPlex HD, 8x8 scans yield significantly more detail and a clearer image compared with the standard AngioPlex system.
My photography team has been impressed. Overall, their satisfaction with new technologies’ speed (particularly useful for patients who tend to blink a lot, which resulted in poor images on slower modalities), rapid montage of images, and improved tracking have led to an improved imaging experience for patients.
Case Presentation
A 50-year-old man with type 2 diabetes mellitus and A1C of 9% came to see me. Wide-field FA imaging highlighted the degree of nonperfusion in the periphery in the capillary bed and the degree of bilateral leakage (Figure 2).
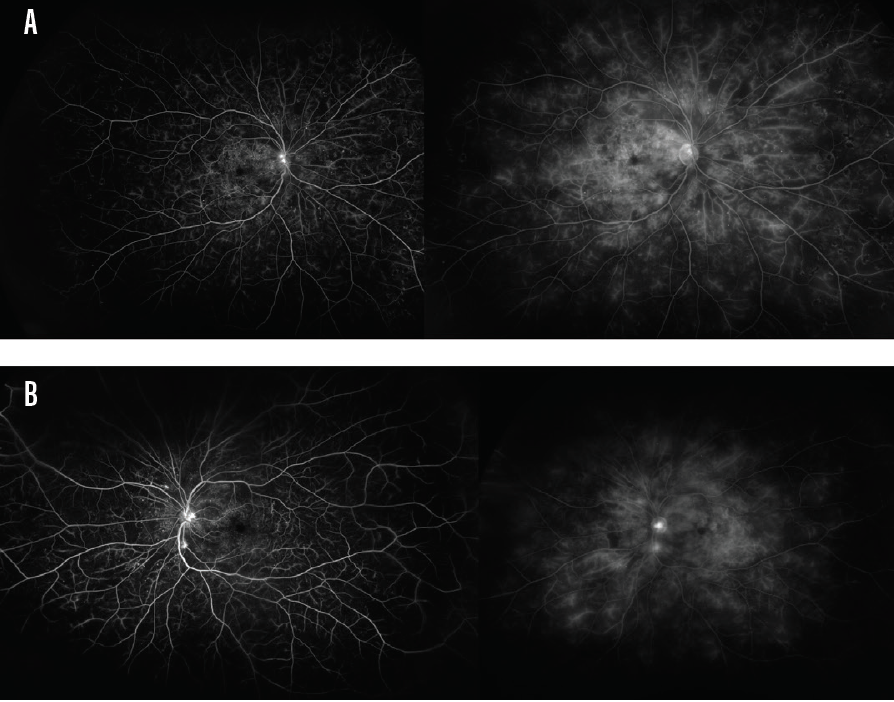
Figure 2. FA imaging results showed that this patient exhibited nonperfusion in the periphery’s capillary bed and leakage in the right eye (A) and left eye (B).
OCTA data added to my understanding of this patient’s condition. An 8x8 montage image of the right eye (Figure 3A) and left eye (Figure 3B) shows the degree of nonperfusion, particularly in the core. The imaging report allowed me to assess the degree to which the perfusion was compromised and the severity of macular edema, and I was able to assess the risk of neovascularization.
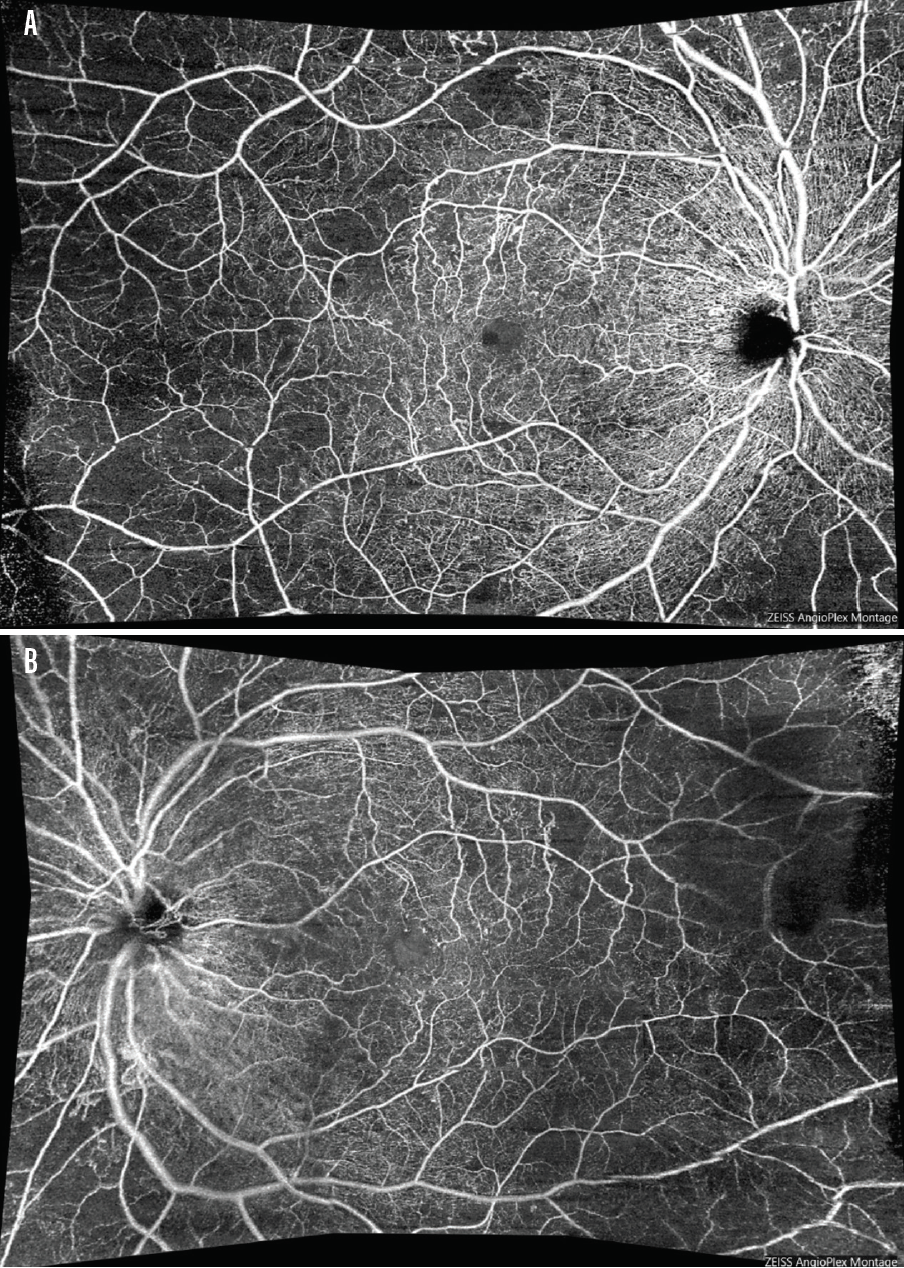
Figure 3. An 8x8 montage image of the right eye (A) and left eye (B) demonstrates severity of nonperfusion, macular edema, and risk of neovascularization.
The choice to initiate anti-VEGF therapy was unaffected by this additional information. However, the ability to track this patient over the course of several visits has improved. By relying on OCTA rather than fundus photography and FA, I can quickly gather information on peripheral perfusion status and neovascularization via a less invasive, faster modality.
It is worth noting that images that some may consider less than adequate actually yield a significant payload of information. The images of patients who blink during imaging, fail to follow instructions, or have comorbidities that interfere with imaging (eg, dry eye disease) still produce data that are useful to the clinician.
UVEITIS
Any imaging I order for patients with uveitis must help me identify patterns, assess disease activity, and tell me something new about the patient’s disease. Sarcoid uveitis may be particularly well suited for swept-source OCTA (SS-OCTA) imaging because the disease manifests in the choroid. ICG angiography (ICGA) is commonly used for this purpose. My team sought to understand if and how OCTA could be used to manage these cases.
Using ImageJ software, participating clinicians at my practice calculated the number of lesions identified in the choroid and choriocapillaris on ICGA and SS-OCTA. We imaged patients on both ICGA and SS-OCTA. On ICGA, 46 (±34) lesions were identified, compared with 52 (±59) lesions on SS-OCTA (P = .744). Of 15 eyes with active disease that were imaged on the same day, 11 had lesions that were identified by both modalities; the remaining 4 eyes did not have lesions identified by either modality.
We measured 31 eyes with active uveitis on SS-OCTA. Flow voids in the choriocapillaris were noted in 22 eyes, and flow voids in the choroid were observed in 16 eyes.
SS-OCTA imaging reports provide data on segmented tissue layers, as opposed to ICGA, which produces a compressed readout. Our team found this useful for tracking lesion activity in patients with active uveitis.
Given that the rates of lesion detection on the two modalities were not significantly different, and given the availability of distinct tissue layer reports, SS-OCTA provides a more advanced imaging option for uveitis management.
CONCLUSION
Advances in imaging have contributed significantly to my clinical decision-making without sacrificing accuracy, efficiency, or patient comfort. As someone who has a high bar for adopting a new imaging modality in my clinic, I can say with confidence that these technologies reach that bar.


