The Goal and Key Questions
Panretinal photocoagulation (PRP) has been the gold standard treatment for diabetic retinopathy (DR) since the results of the Early Treatment Diabetic Retinopathy Study (ETDRS) clinical trial were released in the mid-1980s.1 Intravitreal anti-VEGF injections may yield regression of DR in select patients, thus offering a new option for treatment. Using this medical approach in DR has been likened to slowing a moving train: similar to how steady pressure on a brake mechanism will slow a train, anti-VEGF injections over time offer to slow the disease process, if not stop it in its tracks.2,3 Yet, despite the potential advantages associated with the medical management of DR, there are many unanswered questions surrounding the use of anti-VEGF injections in diabetic eye disease. This roundtable from ASRS 2018 will explore many facets of this new treatment paradigm, including
- What are the current data, and how do those data translate for use in the clinic?
- How are patients selected for therapy?
- What is the optimal timing of doses?
- What treatment endpoints help with decision-making?
- Is it possible to stop treatment at some point, and if so, when and how should that be determined?
- What is the role of laser as adjunctive therapy?
—Rishi P. Singh, MD, Moderator
Reviewing the Data: Protocol S
Rishi P. Singh, MD: Two-year follow-up data from the Protocol S study are now available.2 What are the important findings from this data set?
Nancy Holekamp, MD: The most important outcome from the 2-year data from Protocol S was the improvement in VA, even in the absence of diabetic macular edema (DME).2 We would expect to see VA gains among patients with proliferative DR (PDR) and DME, so this was a somewhat unexpected outcome. The secondary endpoints have not garnered as much attention, but they are equally as important. For example, the rate of vitrectomy was 4% among patients in the anti-VEGF group compared to 15% in the PRP group, and there were fewer cases of retinal detachment, vitreous hemorrhage, iris neovascularization, and neovascular glaucoma. Collectively, these findings seem to suggest that medical therapy arrested the disease process, and as a result, patients experienced fewer complications.
Dr. Singh: Among patients without baseline DME, the anti-VEGF group experienced a +1.3 greater letter gain in VA compared to laser.2 As expected, patients with DME experienced more profound VA gains after medical therapy. The difference in the absence of DME may appear underwhelming.
Tarek S. Hassan, MD: The differences you are highlighting may not be considered what we term “clinically meaningful,” but there is more to the story here. As Dr. Holekamp referred to, anti-VEGF injections yield regression in DR severity, which is supported by reversal in DR severity score in this and other studies.2-4 That regression implies that anti-VEGF injections are impacting the microvasculature of the retina. That is a much different outcome than showing vision gains, and yet it is certainly meaningful to direct therapy to the underlying disease process as well.
Dr. Singh: This trial randomized patients to receive three initial injections and then prn in the anti-VEGF arm. Is that a reasonable way to do this in clinical practice?
Nikolas J.S. London, MD: I start patients on a loading dose, but I prefer to transition to a maintenance injection schedule that is more similar to treat-and-extend than prn. I am interested to see what the outcomes of the Protocol W study (NCT02634333) are to help guide decision-making with respect to timing of doses. For now, I have found that injecting every 3 to 4 months after the loading phase will help prevent recurrent vitreous hemorrhages and other complications.
PROTOCOL S: 5-YEAR RESULTS5,6*
- 69% and 65% of patients in the ranibizumab and panretinal photocoagulation (PRP) group completed 5 years of follow-up (excluding deaths)
- Cumulative mean number of injections over 5 years: 19.2 in ranibizumab group versus 5.4 in PRP group
- Mean change in VA letter score at 5 years was not statistically different: + 3.1 ranibizumab versus + 3.0 PRP
- Mean change in cumulative visual field total point score (-330 vs -537) and rate of developing vision-impairing diabetic macular edema (22% vs 38%) were lower in ranibizumab-treated patients versus PRP-treated patients
- Based on equivalent VA at 5 years of 20/25 in each group, investigators concluded that anti-VEGF and PRP are viable treatment options
*Note: These data were presented at ASRS 2018 after the roundtable discussion occurred. A more robust discussion of these data will be presented in Part Two of this series.
Dr. Singh: Protocol S followed as-needed injections based on whether there was regression of neovascularization. How are you determining the need for subsequent therapy in clinical practice, and are you using imaging to gauge treatment decisions?
Arshad M. Khanani, MD, MA: I believe widefield angiography is the most useful tool for monitoring for DR regression. Color fundus photography is helpful, but fluorescein angiography (FA) will sometimes reveal relevant information about the vasculature and whether the neovascularization is stable. For this reason, I have shifted to performing FA more frequently for these patients.
Dr. Singh: The Protocol S investigators reported that 4% of patients in the anti-VEGF group required a vitrectomy following anti-VEGF injections for fibrosis and tractional detachments. Other studies have noted a similar rate of fibrovascular contraction, although it was lower in Protocol S.7,8 What was your impression of that finding?
Dr. Holekamp: The baseline characteristics in those studies may be important to consider. It seems reasonable that older individuals and those with more severe DR manifestations may be at risk of the crunch phenomenon. We should be cautious about making any conclusions based on comparing data from different clinical trials.
Dr. Singh: As patients being treated with anti-VEGF therapy are followed long-term, what, if anything, are you looking for that might change your mindset?
Dr. Hassan: The biggest thing for me will be whether the treatment effect is sustained or whether we see suggestion of anti-VEGF resistance. The outcomes regarding recurrences of neovascularization will be important. The other thing I am interested to see is if there is any impact from long-term anti-VEGF therapy, such as unusual or unexpected damage to the retinal pigment epithelium. Based on our experience treating other eye diseases with anti-VEGF injections, that is unlikely, but confirmatory data are always helpful.
Dr. London: It will be interesting to see whether treating patients with anti-VEGF therapy is sustainable from a practical standpoint. Historically, these are patients who are younger, potentially of working age, who have a high need for health care resources, and we are asking them to visit for injections several times throughout the year. So, the rate of loss to follow-up may be meaningful to consider. Beyond the ramifications for the study outcomes, this raises questions about what happens to these patients when they come back for treatment. Recent data suggest that 20% to 30% of patients on anti-VEGF therapy are lost to follow-up.9 When those patients return, they tend to have much worse pathology compared to those treated with laser. That is a concern for me, but does that mean we need to rethink how we use laser and how we evaluate the potential for compliance with the treatment protocol? I am not sure we have answers to those questions at the current time.
Reviewing the Data: PANORAMA
Dr. Singh: PANORAMA studied two regimens of aflibercept (Eylea, Regeneron) and observed patients for 52 weeks, with a secondary endpoint at week 100.3 The data at the week 24 endpoint showed a two-step regression of retinopathy in 58% of patients compared with 6% in the sham arm. In contrast to Protocol S, this study enrolled patients with nonproliferative DR (NPDR). The full data might be helpful for deciding how we treat this population of patients. In the meantime, how are you currently treating patients with NPDR?
Panorama Findings3*
Additional findings from the analysis of PANORAMA data at 24 weeks:
- Significant differences in development of vision-threatening complications were noted between sham- and aflibercept-treated patients:
- Development of proliferative diabetic retinopathy or center-involving diabetic macular edema: 25.6% versus 4.5%
- Development of proliferative diabetic retinopathy: 13.5% versus 1.1%
- Development of center-involving diabetic macular edema: 14.4% versus 3.5%
*Note: These data were presented at ASRS 2018 after the roundtable discussion occurred. A more robust discussion of these data will be presented in Part Two of this series.
Dr. Khanani: The paradigm is shifting, but I do not currently treat mild to moderate NPDR with anti-VEGF injections. It will be interesting to see the longer-term data, which might tell us something about the durability and sustainability of this approach. They may also help us identify the best candidates for medical management of DR.
Two points are worth considering in this context:
1. the amount of DR regression largely depends on where you start,10 and
2. 51% of patients with severe NPDR will progress to PDR in 1 year.11
These factors, as well as considerations for the treatment burden, will likely be beneficial for personalizing treatment decisions. There may be a role for treating appropriately selected patients with NPDR to effect a long-term benefit.
Dr. Holekamp: I believe there is or will be a role for treating patients with moderately severe to severe NPDR. The rate of two-step improvement from this study is encouraging, and longer-term data will be interesting. Beyond that, it could also be interesting to quantify the failure to progress.
Dr. Hassan: Failure to progress is a win. That sounds like stalling on train tracks, but it really is not, because we are actually preventing the patient from riding backwards on this disease train. And this may be an apt analogy for another reason in the context of anti-VEGF therapy, because there is potential to help the patient ride towards the goal of disease regression. Treating patients with NPDR with anti-VEGF injections is not simply trying to forestall the inevitable disease progression or maintain the status quo, but rather there is potential to actually improve their underlying retinopathy.
Anti-VEGF: Implications for Clinical Practice
Dr. Singh: What is your current approach to treating patients with PDR, and has your thinking evolved in light of recent data?
Dr. Holekamp: I prefer to start with anti-VEGF injections followed by laser, if needed. My approach has changed in these patients in light of Protocol S and PANORAMA because of the ability to potentially yield regression but also because we can delay the need for laser, which is another way we are helping patients maintain vision with anti-VEGF injections.
Dr. Hassan: Two factors become important to consider for the patient with severe PDR:
1. the severity of the disease and
2. the willingness of the patient to comply with follow-up, which can be difficult to judge.
Often, patients progress to severe PDR because they are not diligent in managing their underlying disease. They may have had diabetes for years, and both of those things are of concern in light of the need for routine monitoring and repeated injections over time. The point was made earlier about patients returning after missing a few appointments with much worse disease, and that is something we will need to consider going forward in terms of whether we use laser alone or in addition to anti-VEGF treatments. We can offer laser or anti-VEGF injections, and how we educate our patients on the importance of compliance will be important.
Dr. London: I have considered creating a contract with the patient, letting them know that, if we start with anti-VEGF injections, here are the potential outcomes, but also that there is some work the patient needs to do. That would put some of the decision on the patient, which I think is appropriate, because, in my experience, outcomes are better when patients are engaged in their own care, especially in diabetes. For us, as retina specialists, the severity of the disease is really the tipping point. In the case of severe PDR, anti-VEGF monotherapy is unlikely to be successful, and either laser or surgery is a possibility. Moderate or earlier NPDR really does not warrant the application of laser; it is really in the early to moderate PDR patient where you have to make a decision. However, for the treatment to be successful, we need the patient to comply with the treatment protocol.
Dr. Khanani: Education starts during the first visit. I like to start the patient with PDR on anti-VEGF injections with instructions to return in 4 to 6 weeks. I explain all the risks and benefits associated with laser and anti-VEGF during the encounter and then give the patient time to absorb that and consider what he or she wants to do going forward.
The Evolving Role of Laser
Dr. Singh: The role of laser in treating DR is evolving. As a result of incorporating anti-VEGF injections for DR and DME, has your use of laser changed?
Dr. Holekamp: The ability to yield regression with anti-VEGF injections2-4 has enabled me to perform less destructive laser even in those cases where the drug alone is not enough to get the result. Laser will likely continue to have a role going forward. For me, even though Protocol S and PANORAMA, together, show that anti-VEGF injections are effective treatments for DR,2,3 we still need longer-term data. On the other hand, by definition, if these people have advanced retinopathy, they likely got that way by being noncompliant. Laser is the current antidote for those two things. As we work through those two issues, we may find ourselves using even less laser.
Dr. Singh: Does anyone incorporate anti-VEGF injections before, during, or after surgery?
Dr. Hassan: I use anti-VEGF agents prior to surgery in only a limited number of cases, but I do think it can be helpful in those select instances. And it can continue to be useful after surgery. The notion that having vitreous surgically removed reduces the efficacy or durability of anti-VEGF injections does not ring true to me. But what we are really discussing here is that anti-VEGF therapy prevents patients from getting to the point where surgery is even indicated.
Dr. Singh: Does anyone use anti-VEGF intraoperatively?
Dr. London: That is not something I do currently, but that is related to the fact that it is difficult to get anti-VEGF injections in the hospital setting I am in. But it is also my impression that there is not much benefit from intraoperative use of anti-VEGF agents. I do perform an injection within the first week postoperatively to reduce the risk of a rebleed.
Dr. Singh: A paper was published a few years ago examining risk factors for postoperative vitreous hemorrhages in patients with PDR.12 The analysis found that the only correlating factor was time since surgery, which probably indicates the number of tractional components dealt with during the surgery.
Practical Considerations
Dr. Singh: How do you counsel your patients when they are started on anti-VEGF therapy with regard to outcomes and follow-up?
Dr. Khanani: I use imaging to show patients why I think it is necessary to start anti-VEGF therapy. I explain that they have a systemic disease that can manifest in the eye. But this is a good opportunity to talk about the importance of systemic control for reducing the risk of losing vision. We also discuss that this approach requires treatment every 4 to 6 weeks initially, with the possibility to extend the interval later but with an indefinite endpoint. It is important to relay the relevant safety information, including the potential for manageable irritation with the actual injection and a very low risk of endophthalmitis. Counseling is also important for managing expectations. I have found that most patients think their vision will improve significantly after the first injection, and they need to be notified in advance about their expectations. Patients with DME and vitreous hemorrhage usually gain vision; others may not notice any improvement, as we are just regressing the PDR to lower the risk of losing vision over time. Over the course of treatment, imaging again becomes useful for educating and showing the patients the impact of treatment on the retina. Overall, I use this opportunity to instill in the patient that this is a team effort, that I can inject a drug that will help treat their DR or DME, but that they need to gain control of their hemoglobin A1C and comply with the treatment schedule.
Dr. Singh: Do you find that patients are aware of the treatment options?
Dr. London: I do not believe patients come in with awareness about what the options are. We have to remember these are typically younger patients, and they have families and busy lives. That may be part of the reason they do not do so well with managing their systemic disease. For that reason, the retina specialist has an opportunity to play an important role in education, not just about the eye disease, but also about the systemic diabetes.
Dr. Khanani: Patients are usually not aware of their treatment options. When we mention that we want to do an injection in the eye, many patients get nervous. I use this opportunity to educate them about the disease, the need for good blood sugar control, and the need for strict follow-up. Compliance is always a challenge with this population, but that partially depends on how involved they are in their own treatment. When patients are more aware of their disease, they are more likely to be engaged.
Dr. London: In the population of patients in my practice in San Diego, patients do not come in with a great deal of knowledge about treatment options, but they do want to learn about their disease and how we can help them.
Dr. Holekamp: There is a growing concept in medicine called “shared decision-making.” I think that is likely to impact very prominently in our management of diabetic patients, precisely because they have a thirst for knowledge. Generally, patients with diabetes want to be a part of the decision-making process.
Dr. Hassan: They need to be involved, because they may have more going on in their lives than the typical age-related macular degeneration (AMD) patients and others in our care. They are more likely to have jobs, families, and younger children, and they may be taking care of their parents who have AMD, for example. They have to be involved in the decision-making process because this is a different population than your 80-year-old AMD patient, and only they can fully understand the life pressures with which they grapple.
Dr. Khanani: How do you see compliance going forward in terms of treating PDR or severe NPDR with anti-VEGF alone?
Dr. Holekamp: By definition, all of these diabetic patients who are in our office with PDR or DME are noncompliant. How do we change behavior? Is it enough to lose your vision to change behavior? Sometimes. Is it enough to tell a patient that getting frequent injections will make an improvement even if they do not get vision improvement? Will that change behavior? That is going to be a challenge.
Dr. Singh: In terms of monitoring patients, is anyone using serial angiography, either FA or OCT angiography? If so, what do you look for on those studies?
Dr. Holekamp: For DR without DME, fundus photographs provide the most compelling information. Showing a patient blood on the retina has an immediate impact, and that degree of alarm is what will drive compliance. Later on, repeat imaging may show improvement, which will encourage continued compliance. For DME, OCT is the most useful imaging we have for patients’ education and for making treatment decisions.
Dr. Khanani: Using anti-VEGF therapy is most successful in the context of personalized medicine. Every patient is different, and many factors, including systemic control, renal status, and health of the cardiovascular system, influence the impact of diabetes on the microvasculature in the eye. For this reason, I repeat an angiogram about every 6 to 12 months to guide treatment, especially if the disease is stable. That usually provides information about the status of proliferative disease and ischemia and helps me understand whether the treatment interval can be extended.
Dr. London: I use OCT angiography and widefield FA. I have found that, in some cases, showing patients macular and peripheral nonperfusion can be motivating. I explain that it is an area where they have lost vessels, and we cannot get them back, and that going forward, we can stabilize or improve what is left if they keep returning for their follow-ups.
Dr. Singh: On the other side of things, is there an endpoint to therapy where injections can be stopped?
Dr. Hassan: We do not know the answer to that question. In my own experience, I have yet to see a patient where all the signs of DR have gone away, but even if they did go away, I would be concerned about it coming back without therapy at some interval, even if it is after quite some time. We need new parameters to understand how we can assess the continued need for treatment.
Dr. London: That really gets to the point of how we are making treatment adjustments and extending the interval.
Dr. Hassan: Unfortunately, we do not have metrics for that, either. I have not gone beyond 3 to 4 months, which possibly raises a question about whether they will actually come back to the office. But when patients ask me what I would do if I were in their position, I tell them I would gladly come into the office every 3 to 4 months to receive an injection if it would help improve the long-term health of my retina and potentially preserve vision indefinitely.
Potential Novel Treatment Approaches
Dr. Singh: There are some new developments lately that may impact treatment of DR in the future. The TIME-2 study investigated a subcutaneous injection of AKB 9778 (Aerpio Pharmaceuticals) alone and in combination with ranibizumab (Lucentis, Genentech).13,14 What are the most important findings from this clinical study?
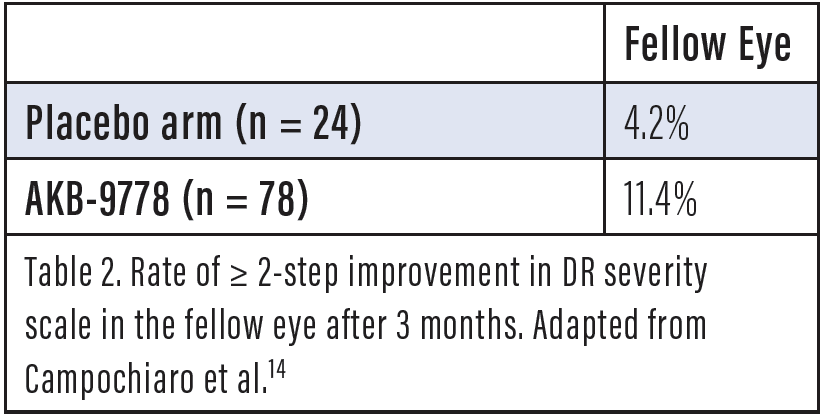
Dr. Khanani: Tie-2 is a transmembrane receptor expressed principally in vascular endothelial cells that promotes stability of the vasculature, but in patients with vascular diseases, including diabetes, Tie-2 is deactivated by increased levels of vascular endothelial protein tyrosine phosphatase or angiopoietin-2 (Ang-2), leading to leakage and pathologic angiogenesis. AKB-9778 is a small molecule that inhibits the intracellular catalytic domain of vascular endothelial protein tyrosine phosphatase. The TIME-2 study enrolled patients with DME to study subcutaneous AKB-9778, 15 mg given twice a day, with a sham intravitreal injection versus subcutaneous AKB-9778 plus a ranibizumab 0.3-mg injection versus a ranibizumab 0.3-mg injection plus subcutaneous placebo. The primary endpoint was change in OCT. In the study, there were no serious adverse events attributable to AKB-9778. As for the results, for the patients in the combination group, there was a statistically significant decrease in central subfoveal thickness, suggesting that restoring Tie-2 activity provided additional benefit compared to ranibizumab 0.3 mg alone (Figure 1).14 Interestingly, in the prespecified DR analysis, the rate of a ≥ 2-step improvement in DR severity scale was about the same among patients who received subcutaneous AKB-9778 alone in the TIME-2 study compared with ranibizumab alone, but importantly without the need for repeated intravitreal injections. Meanwhile, there was a greater rate of fellow eye improvement among those treated with AKB-9778 versus placebo (Tables 1 and 2).14 This is the other benefit of systemic treatment, that you can potentially benefit both eyes. Therefore, AKB-9778 is now being developed as a treatment for DR. The Time-2b study is ongoing and is evaluating the efficacy and safety of subcutaneous AKB-9778 treatment for 48 weeks in patients with moderate to severe NPDR.
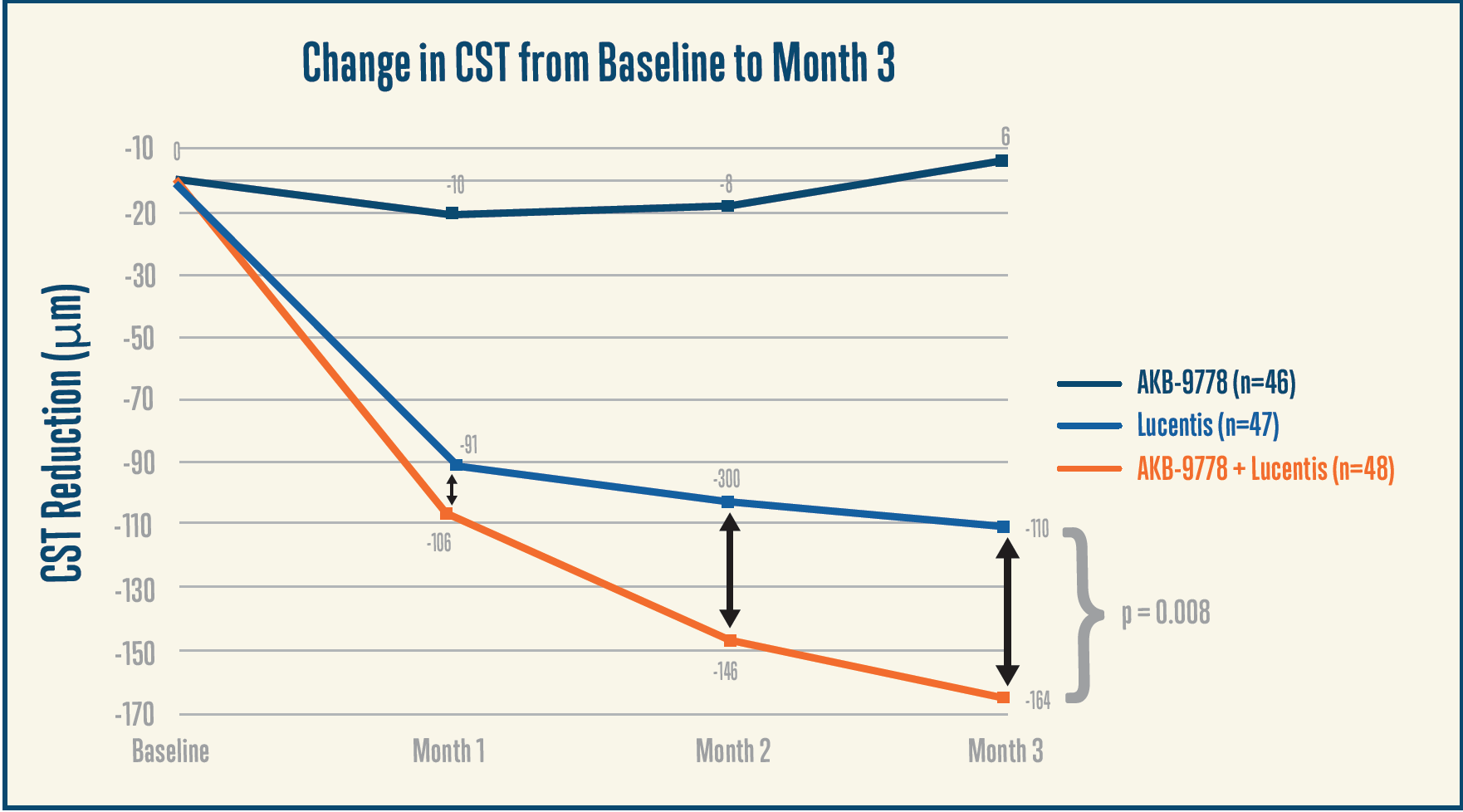
Figure 1. Combination treatment with AKB-9778 and ranibizumab resulted in a greater reduction in central subfoveal thickness (CST) compared to ranibizumab alone, with a continuing separation of the OCT curves at 3 months. Adapted from Campochiaro et al.14

Dr. London: Does using this molecule have potential to reduce anti-VEGF injection burden?
Dr. Khanani: That is unknown as of yet, but as we have discussed here, yielding regression of DR, or at least gaining stability, has every potential to result in an ability to extend the treatment interval.
Dr. Singh: There was no improvement in VA by month 3, but results for OCT outcomes were positive. What is your first impression of these short-term data?
Dr. Holekamp: It is short-term data, and the numbers are small. But it also suggests there is a window of opportunity for combination therapy. The subcutaneous delivery is interesting. Patients with diabetes are used to delivering medications and checking blood subcutaneously, so there is suggestion for good compliance. It is also something they do at home, so that reduces the need to be seen in our clinics. And so, we may reduce the injection burden, but there is still a treatment burden because of the need to monitor patients in the office.
Dr. Singh: Data from the BOULEVARD trial looking at faricimab (formerly RG7716, Genentech) may be worth considering.15 This is a bispecific antibody designed with both an anti-Ang-2 Fab fragment and an anti-VEGF-A fragment attached to an optimized Fc portion. At 24 weeks of follow-up among treatment-naïve patients, there was a +3.6 letter greater improvement among patients in the faricimab plus ranibizumab group compared to the ranibizumab alone group. Treatment was stopped at this point, followed by an observation period to assess for time to regression, which was defined as a 5-letter loss of VA or a 50-µm increase in central serous thickness. At the end of 14 weeks, a higher proportion of treatment-naïve patients maintained vision after the last dose compared to those not treated with faricimab. A similar but less profound effect was noted among individuals who had been previously treated with anti-VEGF injections (Figures 2 and 3).15
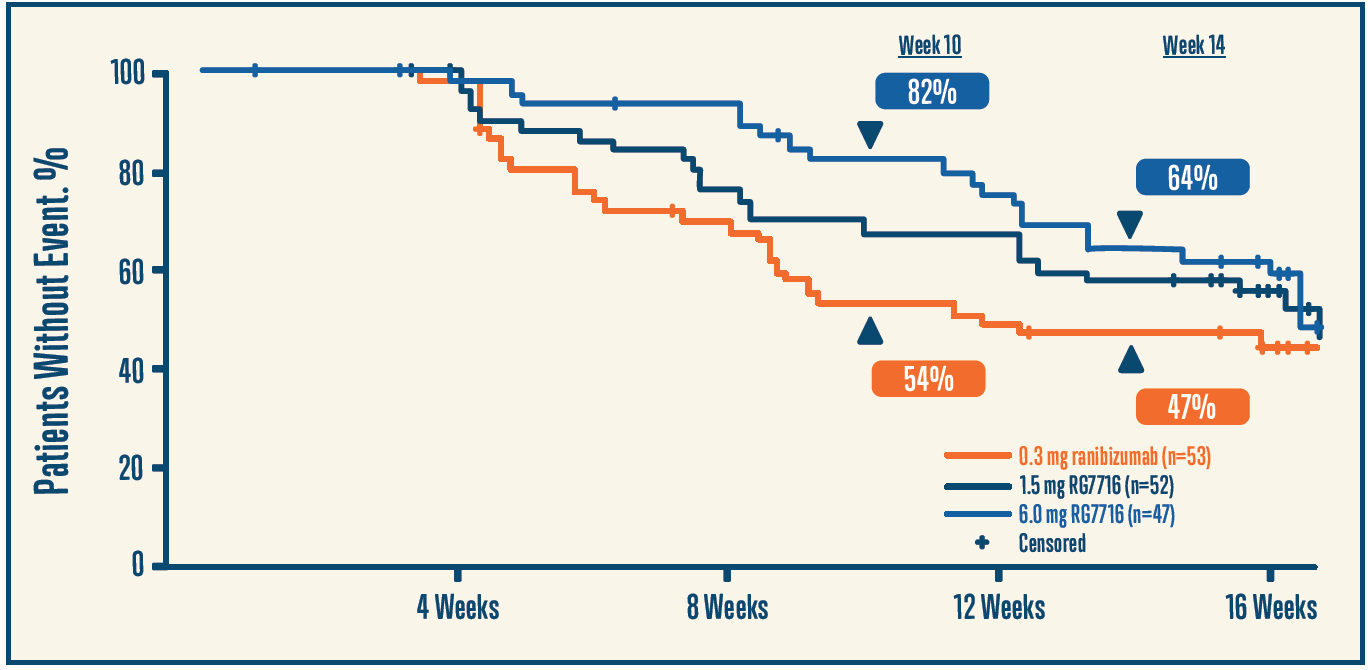
Figure 2. A higher proportion of patients with faricimab maintained vision 12 weeks after the last dose. Adapted from the phase 2 BOULEVARD clinical trial.15
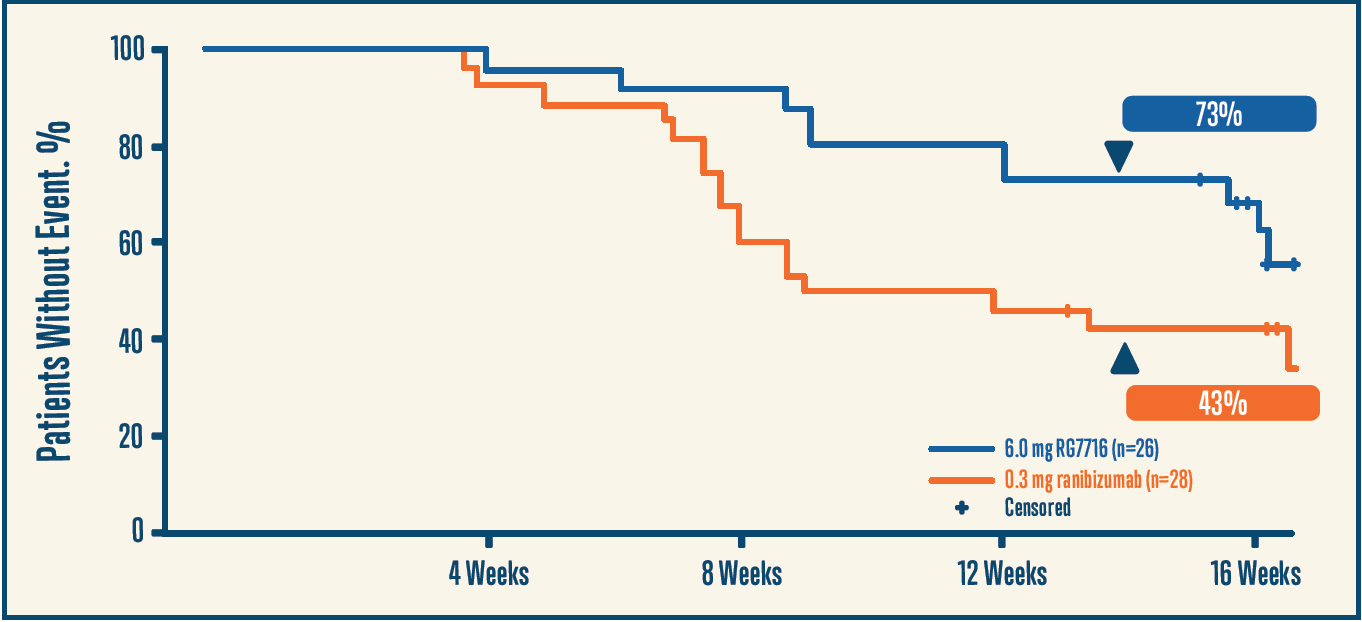
Figure 3. A higher proportion of patients with faricimab maintained disease stability 12 weeks after the last dose. Adapted from the phase 2 BOULEVARD clinical trial.15
Dr. Holekamp: The difference between the treatment-naïve and the treated patients is interesting. It would seem logical that treatment-naïve patients might be earlier in the disease and, therefore, more responsive to both the anti-VEGF and the anti-Ang-2 factors. The durability is notable as well. In the past, we have been treating with anti-VEGF injections without really knowing what the rate of regression would be off therapy, so those data are important.
Dr. Hassan: Treating additional pathways appears to be synergistic with anti-VEGF agents. The pathophysiology of DR and DME is multifactorial, so addressing it in a multipronged approach is rational and likely to be more efficacious, whether that is with this molecule or another way.
Dr. London: Something that is interesting to me is to realize how high the bar has been set for anti-VEGF injections. A +3.6 letter difference with combination versus ranibizumab alone is not a large difference in clinical trials, but that kind of incremental improvement has meaning for patients in the real world.
Dr. Singh: Some additional data from this study were also presented showing a correlation between loss of VA and changes on OCT during the washout period in the treatment-naïve group, but there was almost no connection in the previously treated patients.
Dr. Khanani: That sounds similar to what we saw in the crossover arm in RISE/RIDE.4 Delayed treatment means delayed control of disease, which leads to worse outcomes. Permanent photoreceptor damage is not uncommon among patients who have chronic DME where treatment with anti-VEGF agents was delayed. So, it’s not surprising that, even if the edema improves, VA may not necessarily improve. This is supported by data from DRCR.net Protocol U showing almost no VA benefit with steroids among patients treated long-term with anti-VEGF injections.16 For this molecule, the efficacy in the treatment-naïve patients is impressive, whereas the data for the treatment-experienced population show us the need for early treatment to get the disease under control. If you look at the data for the treatment-naïve patients in the BOULEVARD study, compared to ranibizumab 0.3 mg monthly, patients treated with faricimab 6 mg monthly had better VA, greater reduction in central subfield thickness, higher rates of DR regression, and longer durability during the off-treatment observation period. This clearly shows the benefit of Ang-2 inhibition and stabilizing the Tie-2 pathway in addition to blocking VEGF.
Conclusion
Dr. Singh: Our far-ranging discussion touched on many facets of using anti-VEGF injections for treating DR and DME. There is already one anti-VEGF agent approved for use in patients with DR with or without DME, ranibizumab, while a second, aflibercept, is currently being reviewed by the US FDA for this same indication—with an expected action date by May 2019.17 The data we have so far show us the potential to yield regression with disease severity. Using anti-VEGF therapy in this manner has been described like slowing a moving train. We cannot stop the disease with a single injection, but continued therapy over time slows or stops progression.2,3 This is, indeed, a whole new paradigm for treatment of DR with potential to intervene earlier. While we are at a stage when we do not have precise parameters for selecting appropriate patients and for making decisions about when to treat and when to adjust therapy, this is also not new territory for retina specialists: it was not that long ago that anti-VEGF agents were introduced into DME, and that rapidly became the standard of care for center-involving DME after we found answers to similar questions. What are some early-term impressions with this new treatment approach?
Dr. Khanani: PRP was the only treatment for decades, but we now have anti-VEGF agents as an option for our patients with DR. The efficacy of anti-VEGF agents in DR has been supported by several clinical trials. As far as anti-VEGF therapy is concerned, there is strong rationale for use in patients with PDR, with a role for adjunctive laser. In terms of severe NPDR and earlier, there are very good data to support treatment, but in the real world, anti-VEGF use is still discretionary, and the decision needs to be individualized. As we gather more data, that may well change—and I would include in this the new studies on novel molecules addressing additional pathways in DR and DME pathogenesis.
Dr. Holekamp: The indication for using anti-VEGF agents in DR is an evolution rather than a revolution but nonetheless important. We do not want to be treating patients with DR in 5 or 10 years the same way we are now. Adding new approaches offers to improve outcomes, both anatomically and with regard to visual outcomes. There is some complexity, but as retina specialists, we should stay dynamic as the treatment approach evolves.
Dr. Hassan: The retina community now has experience from clinical trials and personal use with anti-VEGF agents in DR and DME, and this allows us to help our patients make truly informed choices about their treatment. Laser has proven highly successful, and some patients may opt for this approach. But in the interest of full disclosure, we should be educating patients about this alternative option. The data give us the confidence to educate patients about the potential for improved vision and stability of disease with repeat injections, and losing peripheral vision after laser is not something they have to risk. From a practical standpoint, it will be a matter of how it is integrated into one’s practice, although there is likely relevant experience in delivering these agents in patients with AMD and retinal vein occlusions from which to draw upon.
Dr. London: As retina specialists, we are not necessarily used to treating prophylactically, and we are much more attuned to addressing a problem after it occurs. Starting treatment after conversion to wet AMD is one example. With DR, we are talking about initiating treatment to prevent worsening of disease. That is going to take a slightly different mindset to be successful, especially for patients with NPDR who may not be symptomatic. Returning to the Protocol S data, about 25% of patients in the PRP group developed a vitreous hemorrhage.2 Essentially, if you are waiting for PDR to occur, you are willing to accept the risk of vitreous hemorrhage, whereas we saw in RISE/RIDE that the rate of vitreous hemorrhage is lower after anti-VEGF treatment.4 We should be cautious about comparing across studies, but I think this highlights that we owe it to patients to explain the differences between the treatment options. When I have done so in a very honest manner with my patients, I have found the vast majority would prefer to reverse their disease and would prefer to preserve their peripheral retina, their night vision, and their visual field.
Dr. Singh: Thank you, everyone, for your participation.
1. Early Treatment Diabetic Retinopathy Study Research Group. Early photocoagulation for diabetic retinopathy. ETDRS report number 9. Ophthalmology. 1991;98(5 Suppl):766-785.
2. Gross JG, Glassman AR, Jampol LM, et al; Writing Committee for the Diabetic Retinopathy Clinical Research Network. Panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: a randomized clinical trial. JAMA. 2015;314(20):2137-2146.
3. Wykoff C. Intravitreal aflibercept for moderately severe to severe nonproliferative diabetic retinopathy (NPDR): the phase 3 PANORAMA study. Presented at the ASRS Annual Meeting, July 20-25, 2018; Vancouver, BC.
4. Nguyen QD, Brown DM, Marcus DM, et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119:789-801.
5. Gross JG, Glassman AR, Liu D, et al; Diabetic Retinopathy Clinical Research Network. Five-year outcomes of panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: a randomized clinical trial. JAMA Ophthalmol. 2018. [Epub ahead of print]
6. DRCR.net. Five-year outcomes of panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy. Available at: http://drcrnet.jaeb.org/ViewPage.aspx?PageName=Presentations. Accessed September 26, 2018.
7. Avery RL, Pearlman J, Pieramici DJ, et al. Intravitreal bevacizumab (Avastin) in the treatment of proliferative diabetic retinopathy. Ophthalmology. 2006;113(10):1695-1705.
8. Larrañaga-Fragoso P, Peralta J, Bravo-Ljubetic L, et al. Intravitreal bevacizumab for zone II retinopathy of prematurity. J Pediatr Ophthalmol Strabismus. 2016;53(6):375-382.
9. Obeid A, Su D, Patel SN, et al. Outcomes of eyes lost to follow-up with proliferative diabetic retinopathy that received panretinal photocoagulation vs intravitreal anti-vascular endothelial growth factor. Ophthalmology. 2018. [Epub ahead of print]
10. Dhoot DS, Baker K, Saroj N, et al. Baseline factors affecting changes in diabetic retinopathy severity scale score after intravitreal aflibercept or laser for diabetic macular edema: post hoc analyses from VISTA and VIVID. Ophthalmology. 2018;125(1):51-56.
11. Early Treatment Diabetic Retinopathy Study Research Group. Fundus photographic risk factors for progression of diabetic retinopathy. ETDRS report number 12. Ophthalmology. 1991;98(5 Suppl):823-833.
12. Ahn J, Woo SJ, Chung H, et al. The effect of adjunctive intravitreal bevacizumab for preventing postvitrectomy hemorrhage in proliferative diabetic retinopathy. Ophthalmology. 2011;118(11):2218-2226.
13. Khanani A. AKB-9778 in the treatment of diabetic macular edema: results from the TIME-2 study. Presented at the ASRS Annual Meeting, August 9-14, 2016; San Francisco, CA.
14. Campochiaro PA, Khanani A, Singer M, et al; TIME-2 Study Group. Enhanced benefit in diabetic macular edema from AKB-9778 Tie2 activation combined with vascular endothelial growth factor suppression. Ophthalmology. 2016;123(8):1722-1730.
15. Singh R. Anti-VEGF/anti-angiopoietin-2 bispecific antibody RG7716 in diabetic macular edema: 36-week results from the phase 2 BOULEVARD clinical trial. Presented at the ASRS Annual Meeting, July 20-25, 2018; Vancouver, BC.
16. Maturi RK, Glassman AR, Liu D, et al; Diabetic Retinopathy Clinical Research Network. Effect of adding dexamethasone to continued ranibizumab treatment in patients with persistent diabetic macular edema: a DRCR network phase 2 randomized clinical trial. JAMA Ophthalmol. 2018;136(1):29-38.
17. Regeneron. FDA to review EYLEA® (aflibercept) injection for the treatment of diabetic retinopathy. Available at: https://investor.regeneron.com/news-releases/news-release-details/fda-review-eylea-aflibercept-injection-treatment-diabetic. Accessed September 26, 2018.









